| CellSeed (7776) |
|
||||||||
Company |
CellSeed Inc. |
||
Code No. |
7776 |
||
Exchange |
JASDAQ |
||
Industry |
Regenerative Medicine |
||
President |
Yukio Hasegawa |
||
HQ Address |
R-Bldg., Shinjuku, 33-8, Wakamatsu-cho, Shinjuku-ku, Tokyo, Japan |
||
Year-end |
December |
||
URL |
|||
* Stock price as of closing on Feb. 22, 2012. Number of shares at the end of the most recent quarter excluding treasury shares.
ROE, BPS are based on actual results as of the previous term end. |
||||||||||||||||||||||||
|
|
* Estimates are those of the Company.
Mail: CellSeed@cyber-ir.co.jp |
| Key Points |
 |
| Company Overview |
|
Cell Sheet Regenerative Medicine Business
Cell sheet regenerative medical products (Cell sheets) and various applications of these products are sold in this business. Currently CellSeed is promoting joint research and development of five different regenerative medical product pipelines (New pharmaceutical product candidates) with collaborative research partners.
Regenerative Medicine Support Business
CellSeed develops and manufactures temperature responsive cell cultureware (CellSeed is the only manufacturer of these products in the world) and various applications of this product (Some of the manufacturing processes that require large capital investments are outsourced.), that are the fundamental tool in cell sheet regenerative medicine. These products are provided to universities, research institutions and other institutions around the world. This business is of high strategic importance in developing partners in the cell sheet regenerative medicine business, in addition to its main purpose of generating earnings.
 |
| Fiscal Year December 2011 Earnings Results |
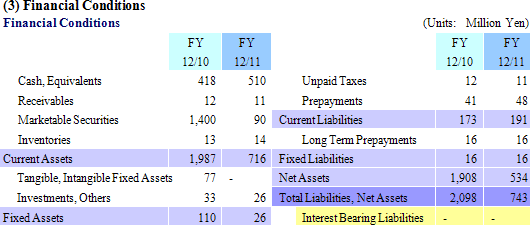
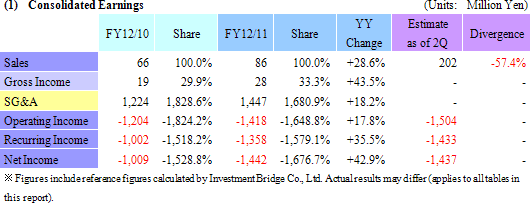 Recurring Loss of ¥1.358 Billion In Line with Expectations
Sales of ¥86 million were booked in the regenerative medicine support business. Sales of both temperature responsive cell cultureware and products in overseas markets exceeded plans. And while cell sheet regenerative medicine business was unable to realize any sales, corneal regeneration epithelial cell sheets entered the final phase (Medical approval process) during the second quarter (April to June) in Europe prior to its market launch, and other product pipelines entered the human clinical research stage.With regards to profits, efforts to reduce expenses achieved some success (SG&A expenses excluding research and development fell from ¥615 to ¥579 million), but research and development expenses rose from ¥609 to ¥867 million (About ¥200 million in development expenses for corneal regeneration epithelial cell sheets in Europe have been postponed until fiscal year December 2012) and contributed to an expansion in operating loss. Declines in subsidies (Fell from ¥226 to ¥55 million) led to a deterioration in non-operating income, and a loss arising from impairment accounting of ¥72 million also contributed to an extraordinary loss of ¥79 million (Extraordinary loss in the previous term amounted to ¥3 million). Both sales and losses in fiscal year December 2011 were in line with estimates revised in December. However when compared with estimates announced at the end of the first half, delays in the lump sum payment of US$1.50 million (\127 million at the exchange rate in existence at the time of the signing of the contract) by Emmaus Medical, Inc. for the commercialization of corneal regeneration epithelial cell sheets as part of the cell sheet regenerative medicine business in the United States until fiscal year December 2012 were the main factor behind the large shortfall in sales. At the same time, losses fell below expectations due to delays in realization of some ¥200 million in research and development expenses for corneal regeneration epithelial cell sheet in Europe until fiscal year December 2012. 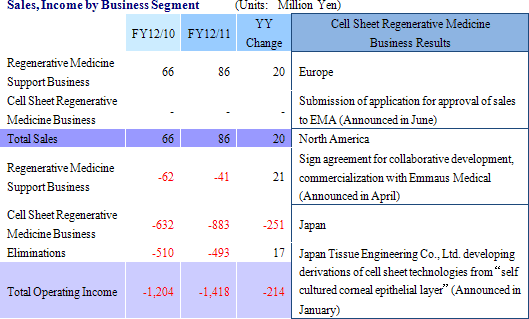 (2) Progress in the Cell Sheet Regenerative Medicine Business
① Corneal Epithelial Cell Sheet in Europe
An approval application for commercial launch was submitted to the European Medicines Agency (EMA) on May 31, 2011 and confirmation of its acceptance was received on June 7. The application was found to be filled out satisfactorily and the EMA is currently conducting a screening of the application. Furthermore, regenerative medicine is categorized in non-traditional "Advanced Therapy Medicinal Product" (ATMP) grouping (Corneal Regeneration Epithelial Cell Sheet has been designated as an ATMP) according to European pharmaceutical regulations. Therefore pharmaceutical screening of this treatment has been conducted by the EMA, which encompasses pharmaceutical issues for the overall EU region, and not by the pharmaceutical screening process of the individual countries belonging to the EU. Therefore pharmaceutical products that have undergone medical screening process of the EMA and acquire marketing approval of the EC will be recognized for sales to over 500 million people in the 30 member countries of the EU market (27 countries belonging to the EU and 3 countries falling under EEA/EFTA).Furthermore, Helmut Hasibeder, the former President of Takeda Pharmaceutical Company Limited Group’s Austrian subsidiary, has been assigned to the responsibility of marketing and sales in the European market. Consequently the sales collaboration agreement with Clonmel Healthcare Limited has been dissolved. At the same time, compassionate use of products in France (Provision of pharmaceutical products which have yet to receive medical approval to patients in accordance with the guidelines in various European countries) has been stopped due to the reinforcement of regulations (To be explained later). ② Other Product Pipelines
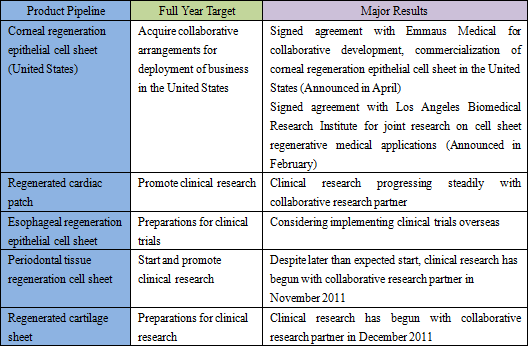
Research and development on product pipelines other than the corneal regeneration epithelial cell sheet in Europe are steadily progressing, and five product pipelines, including corneal regeneration epithelial cell sheet in the United States, have all entered human clinical research phase.
   (4) Overview of Patents Acquired in Fiscal Year December 2011
The main businesses of CellSeed can be divided between the three main tasks of "cell gathering and separation," "construction of tissue and organs using cell sheet engineering," and "transplant therapies." During fiscal year December 2011, six patents (Four within Japan, two in Europe) were acquired in the realm of the first two processes of "cell gathering and separation" and "construction of tissue and organs using cell sheet engineering," including patents for a new material "high density cell array substrate" necessary for the "cell gathering and separation" process. Another two more patents were acquired in the "transplant therapies" process, including one for the reconstruction of "cancer tissue model construction technology" using cell sheet (Cell sheet usage evaluation system), bringing the total number of patents acquired to eight. Details of the acquired patents are provided below.
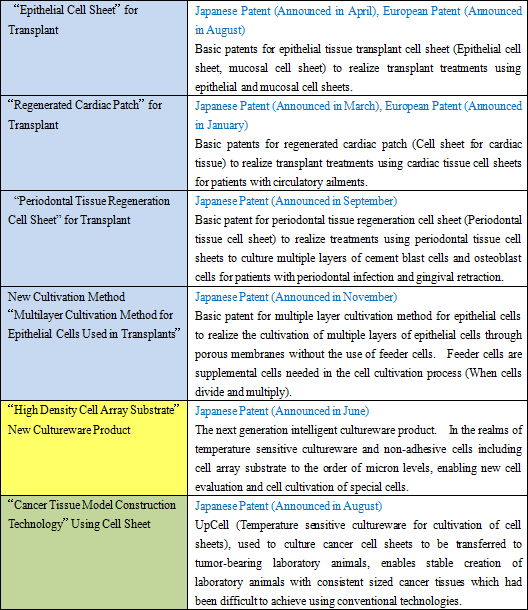 Moreover, cell sheet used in "cancer tissue model construction technology" allow specific sized cancer cells to be used in the realm of cancer model animals (Specific sized cancer cells can be created and placed in specific locations). |
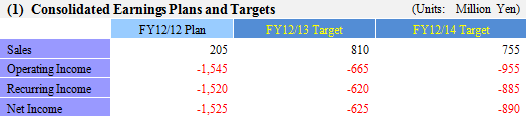
| Three Year Business Plan (From FY12/12 to FY12/14) |
 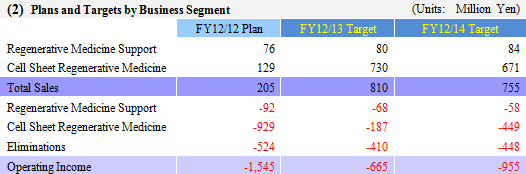 Regenerative Medicine Support Business
Continue to promote research and development for new products in addition to efforts to increase sales through fortification of the product lineup and marketing promotion activities.
Cell Sheet Regenerative Medicine Business
During fiscal year December 2012, CellSeed expects to see revenues from a lump sum payment to be received from Emmaus Medical for the commercialization of the corneal regeneration epithelial cell sheet business in the United States, in addition to the launch of compassionate usage of corneal regeneration epithelial cell sheets in Europe. However with regards to the corneal regeneration epithelial cell sheet business in Europe, the need to provide additional data in addition to the intermediate term data (Short term data) of clinical trials being conducted at other facilities is expected to lead to delays in the start of sales until fiscal year December 2013. Aside from this, regenerated cardiac patch is expected to begin clinical trials and preparations for clinical trials of esophageal regeneration epithelial cell sheet (Clinical trial is expected to begin during fiscal year December 2013) are expected to begin during the current term. Periodontal tissue regeneration cell sheet (Clinical research is expected to be completed in fiscal year December 2014) and regenerated cartilage sheet will continue to undergo clinical research.During fiscal year December 2013, a lump sum payment for joint research and development for regenerated cardiac patch is expected to be acquired in addition to revenues arising from the approval for sale (Conditional approval, which may require annual reporting) of corneal regeneration epithelial cell sheet in Europe. During fiscal year December 2014, sales are expected to decline due to the disappearance of lump sum payments. Moreover the compassionate use system allows for pre-approved drugs to be provided to human patients on a paid basis in the major countries throughout Europe.  (3) Near Term Financial Strategy
During fiscal year December 2011, capital needs required CellSeed to liquidate its marketable securities as well as securing its capital requirements for fiscal year December 2012 forward through the use of stock options (Total of Nomura Equity Line: ¥1.000 billion, of which ¥900 million remains unused).During fiscal year December 2012, in addition to Nomura's Equity Line mentioned above, a lump sum payment from Emmaus Medical (Two payments totaling US$10 million) is also expected. And during fiscal year December 2013, a lump sum payment for joint research and development of regenerated cardiac patch (\500 million) is expected to be received, in addition to funds from expansions in collaborative partnerships and public and other subsidies. Of the above mentioned sources of funds, Nomura Equity Line, and lump sum fees from Emmaus Medical for collaborative work and for joint research and development of regenerated cardiac patch have a high probability of being booked. Combined with the total cash on hand currently, these funds will bring the total cash available to CellSeed to approximately ¥2.800 billion, which should be adequate to meet the Company's cash needs through fiscal year December 2013, as well as covering the bulk of the cash needs of fiscal year December 2014. |
| Highlights of Fiscal Year December 2011 Earnings and Business Plans Going Forward |
|
With regards to the revision to the compassionate use plans described earlier, these revisions can be attributed to conditions that existed within France. These types of unforeseen factors are not unusual for any newly started business. And CellSeed's conservative outlook for contributions from compassionate use allowed the impact of this revision to be negligible. Furthermore there are no problems for the corneal regeneration epithelial cell sheet application and its business potential is still viable. With regards to delays in commercialization of the corneal regeneration epithelial cell sheet business in Europe (Due to delays in acquisition of medical approval), "reproducibility" appears to be the main issue. Put simply, the clinical trials, which were successful, for the corneal regeneration epithelial cell sheet were conducted by a well known doctor at Lyon National Hospital, and the regulatory authorities want to see if the same successful results can be achieved at other facilities and by different doctors. In the normal process for medical approval, CellSeed has been questioned by the regulatory authorities after it has submitted various documents, and from these interviews the Company felt that there were lingering questions with regards to the issue of "reproducibility." Therefore results from further clinical trials at other facilities are expected to be submitted to the regulatory authorities (Clinical trials at a smaller number of facilities requires more time as data from more samples need to be taken, whereas clinical trials at numerous facilities requires less time because data from a lesser number of samples at each facility is needed.) to answer these questions. But because the screening process will be halted until the questions are answered, the total screening time for the approval is expected to lengthen. In other words, this delay does not reflect a fundamental change in the outlook for the approval but it is necessary to gather more data due to the regulatory authority's conservative approach to regenerative medical treatments. Furthermore the delay in the lump sum payment for the commercialization of the corneal regeneration epithelial cell sheets in the United States does not reflect any long term issues for this business. CellSeed had anticipated a lump sum payment of US$1.50 million to be received for technology transfer to Emmaus Medical and for the documentation for medical approval in Europe, but the need to provide data from transplants performed upon laboratory animals has arisen. Therefore the lump sum payment has been delayed until after the data can be prepared, but transplant experiments upon laboratory animals have already been completed (Model animal creation has been completed and they are currently undergoing observation.), and CellSeed expects payment of the fees to be made at an early stage. Furthermore with regards to the remaining US$8.50 million, while some slight delays are expected due to the need to exchange various information about manufacturing facilities before manufacturing can be started, payment is expected to be received within fiscal year December 2012 (This fee has not been included in plans for fiscal year December 2012). In addition to these issues, the timing of a lump sum payment arising from joint research for regenerated cardiac patch has yet to be to be determined. |
| Conclusions |
|
Disclaimer
This report is intended solely for information purposes, and is not intended as a solicitation to invest in the shares of this company. The information and opinions contained within this report are based on data made publicly available by the Company, and comes from sources that we judge to be reliable. However we cannot guarantee the accuracy or completeness of the data. This report is not a guarantee of the accuracy, completeness or validity of said information and or opinions, nor do we bear any responsibility for the same. All rights pertaining to this report belong to Investment Bridge Co., Ltd., which may change the contents thereof at any time without prior notice. All investment decisions are the responsibility of the individual and should be made only after proper consideration.Copyright(C) 2012, All Rights Reserved by Investment Bridge Co., Ltd. |



