| CellSeed (7776) |
|
||||||||
Company |
CellSeed Inc. |
||
Code No. |
7776 |
||
Exchange |
JASDAQ |
||
Industry |
Regenerative Medicine |
||
President |
Yukio Hasegawa |
||
HQ Address |
R-Bldg., Shinjuku 33-8, Wakamatsu-cho, Shinjuku-ku, Tokyo |
||
Year-end |
December |
||
URL |
|||
* Stock price as of closing on 2011/8/18. Number of shares at the end of the most recent quarter excluding treasury shares.
ROE, BPS are based on actual results as of the previous term end. |
||||||||||||||||||||||||
|
|
* Estimates are those of the Company. Data for fiscal year December 2007 to 2008 are non-consolidated.
A stock split of 100-for-1 was undertaken in October 2010. Mail: CellSeed@cyber-ir.co.jp |
|
| Key Points |
 |
| Company Overview |
|
Regenerative Medicine Support Business
CellSeed develops and manufactures temperature responsive cell cultureware and various applications of this product (Some of the manufacturing processes that require large capital investments are outsourced.) that are the fundamental tool in cell sheet regenerative medicine. These products are provided to universities, research institutions and other institutions around the world. This business is of high strategic importance in developing partners in the cell sheet regenerative medicine business, in addition to its main purpose of generating earnings.
Cell Sheet Regenerative Medicine Business
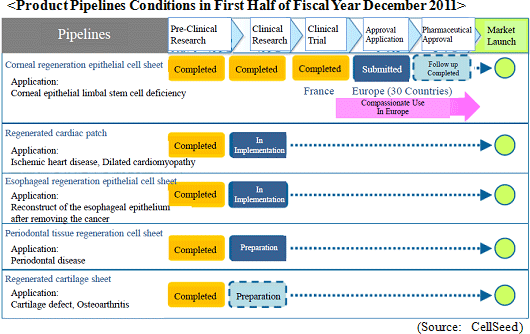
Cell sheet regenerative medical products (Cell sheets) and various applications of these products are sold in this business. Currently CellSeed is promoting joint research and development of five different regenerative medical product pipelines (New pharmaceutical product candidates) with collaborative research partners.
 |
| First Half of Fiscal Year December 2011 Earnings Results |
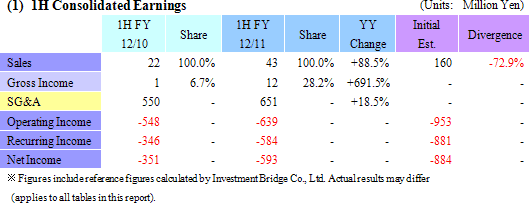 Results In Line with Revised Estimates Announced on July 15
Sales of ¥43 million were recorded. During the first half, sales of temperature responsive cell cultureware and the new cell tight junction real monitoring system "cellZscope" product launched last summer exceeded initial expectations held at the start of the term. At the same time, while no sales were recorded in the cell sheet regenerative medicine business segment, the approval application for the commercial launch of corneal regeneration epithelial cell sheet applications was accepted in June by the European Medicines Agency (EMA) and represents a positive step towards realizing future earnings. However, current conditions are causing research and development to become a heavy burden upon selling, general and administrative (SG&A) costs and contributed to the operating loss of ¥639 million. The operating loss increased from the previous first half due in part to research and development expenses, which account for more than half of SG&A costs and rose by ¥117 million to ¥380 million from ¥263 million in the previous first half. At the same time, reductions in various expenses allowed SG&A costs, excluding research and development expenses, to actually decline from ¥286 to ¥271 million, and allowed the operating loss to fall significantly short of the highly conservative revised estimates announced along with first quarter earnings results. Furthermore, a reason why sales fell below estimates by such a large margin was the delay in a lump sum payment of US$1.5 million (¥127 million) by Emmaus Medical Inc. during the first half, which is now expected to be paid in the second half (Technology licensing work expected to be completed during the second half relating to the contract for the commercialization and joint development of corneal regeneration epithelial cell sheets in the United States.).  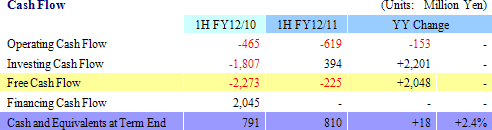  There is currently no effective treatment for patients with severe ischemic heart disease and dilated cardiomyopathy. Therefore regenerated cardiac patches are created from myoblast cells taken from the thighs of patients and are grafted back to the patients affected hearts. Regenerated cardiac patches have been used to treat 10 patients thus far. Esophageal regeneration epithelial cell sheets are being developed as a regenerative medicine product pipeline used to reconstruct the esophageal area of patients who have had cancer infected parts of their esophagus removed, and to restrain or prevent esophageal narrowing (Condition where a portion of the esophagus narrows, causing a blockage when swallowing foods) after cancerous portions are removed. Cell sheets (Esophageal regeneration epithelial cell sheet) created from oral cavity mucosal epithelial cells have been grafted to patients who have had cancerous portions of their esophagus removed to restrain inflammatory response at an early stage and to prevent esophageal narrowing in 10 patients. During the current term, CellSeed is expected to begin preparations for clinical trials. Periodontal tissue regeneration cell sheet is being developed as a product pipeline to treat patients who have lost periodontal tissue due to periodontal diseases and those who have periodontal diseases indications. Cell sheets created from cells of the periodontal ligament (Periodontal tissue regeneration cell sheet), which are essential in regeneration of teeth and gum tissue, are grafted to patients suffering from periodontal diseases to reconstruct lost periodontal tissue. During the current term, clinical research is expected to begin and the selection process of patients who will be the subjects of clinical research is currently being conducted. Regenerated cartilage sheet cultivated created from the patient' s own cartilage cells to be grafted to the patient is being developed as a product pipeline for use as a treatment for cartilage defect and osteoarthritis. Currently facilitation of data and documents (The Ministry of Health, Labor and Welfare is conducting deliberations for the start of clinical research) for the start of clinical research is being conducted. (3) Topics: Japan-US-EU Tri-Polar, Simultaneous Research and Development Structure Promoted, Various Patents Established for Corneal Regeneration Epithelial Cell Sheet
[1] Japan-US-EU Tri-Polar, Simultaneous Research and Development Structure for Corneal Regeneration Epithelial Cell Sheet
A tri-polar and simultaneous research and development structure is being created in Japan, the United States and Europe for the corneal regeneration epithelial cell sheet business. Also commercial marketing approval application has been accepted by the EMA in June, and a contract for joint development and commercialization of this product has been signed with Emmaus Medical Inc. of the United States in April. In addition, Japan Tissue Engineering Co., Ltd. is developing "Self Cultured Corneal Epithelial Layer" in Japan and has participated in cell sheet engineering.
[1]-1 Europe: Marketing Launch Approval Application Submitted to the European Medicines Agency (EMA)
The approval application for the commercial launch of corneal regeneration epithelial cell sheet applications was accepted in June by the European Medicines Agency (EMA). Based upon preliminary discussions, the EMA has provided positive scientific advice and the approval process is expected to proceed smoothly. However the period of time required for approval varies from case to case. The approval period is also influenced by the time required to respond to questions asked, but approval is expected to be granted by the end of fiscal year December 2012 at the very latest.Furthermore, regenerative medicine is categorized in non-traditional "Advance Therapy Medicinal Product" (ATMP) grouping (Corneal Regeneration Epithelial Cell Sheet has been designated as a ATMP) according to European pharmaceutical regulations. Therefore pharmaceutical screening of this treatment will be conducted by the EMA, which encompasses pharmaceutical issues for the overall EU region, and not by the pharmaceutical screening process of the individual countries belonging to the EU. The EMA pharmaceutical screening process is designed to grant final marketing approval of the European Commission to candidate pharmaceuticals which have been proven to be safe and effective in the treatment of corresponding diseases, and this marketing approval of the European Commission is recognized as an official pharmaceutical approval in the 30 countries belonging to the EU member nations. Therefore in the event that corneal regeneration epithelial cell sheet receives European Commission marketing approval, sales will be started in the 30 member countries of the EU market where over 500 million people reside (The pharmaceutical pricing needs to be determined in each respective country.). [1]-2 United States: Collaboration with Emmaus Medical Inc., Join Research Agreement Signed with LA Bio Med
In April, CellSeed signed agreements with Emmaus Medical Inc. (Torrance, California, U.S.) for "basic agreement for joint research and development" and "agreement for joint research and development and commercialization of corneal regeneration epithelial cell sheet technologies." Through these agreements, CellSeed will be able to put capital resources, pharmaceutical product research and development knowhow, and network of personal contacts of Emmaus in the United States to work to help establish its operational base in the United States and accelerate its business deployment there.Two agreements signed for the comprehensive research and development of cell sheet regenerative medicine in the United States include the "basic agreement for joint research and development" and an individual agreement called the "agreement for joint research and development and commercialization of corneal epithelial cell sheet technologies." The latter individual agreement entails a lump sum payment of US$1.5 million and payment (To be recorded as sales) is expected to be made during the second half of the current term along with the completion of the technology provision task and has been included in full year estimates. And while an US$8.5 million lump sum payment is expected to be received for the "basic agreement for joint research and development" within the current term, CellSeed is currently considering how to treat this payment in terms of its accounting practices. Emmaus Medical was established as a drug discovery venture in California in 2003 by Yutaka Niihara, PhD. (Professor at UCLA Medical School) and others with the mission of developing applications using L-glutamine (A type of amino acid found commonly in muscle tissue that was discovered by Yutaka Niihara.) that is effective in the treatment of difficult diseases. Applications of L-glutamine effective in the treatment of short bowel syndrome (Causing deficiency in absorption due to removal of parts of the small intestine.) have already been launched into the market, and clinical trials of applications in the treatment of drepanocyte disease (Anemia caused by hereditary conditions.) are currently being conducted (Currently in phase 3 clinical trial). Emmaus Medical is gathering the attention of the medical community not only in the United States, but in Japan and other parts of the world as well. Also Henry A. McKinnell Jr., PhD., who was formerly the CEO of Pfizer Inc., is also an external director. Emmaus Medical was capitalized at US$13.8 million and is a shareholder of CellSeed (As of end June 2011). ・ Joint Research And Development Agreement with LA Bio Med
In February, CellSeed entered into an agreement (Covering a two year period) to conduct joint research in regenerative medical applications using cell sheet regenerative engineering that are a fundamental part of regenerative medical technologies with Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center (LA Biomed), one of the largest non-profit medical institutions in the United States.LA Biomed was established in 1952 in Torrance, California as a non-profit medical institution and employs over 150 research staff who conduct research projects designed to develop new treatments for patients suffering with cancer, hereditary diseases, infectious diseases and other diseases (Research on over 1,000 research themes is being conducted.). In addition to close technological collaboration with the David Geffen School of Medicine at UCLA, LA Biomed also trains young technicians, provides immunization programs, provides instruction on infant nutrition, and maintains a goal of contributing to the community healthcare. Thereby LA Biomed has established a position as the leading institution in the field of medicine and medical care in the South Los Angeles area. A team led by Yutaka Niihara, PhD. (Professor at UCLA Medical School) is undertaking joint research, and CellSeed hopes to use this joint research to promote the commercial launch of its corneal regeneration epithelial cell sheet, which is currently undergoing preparation for commercial launch in Europe, in the United States at an early stage as well. CellSeed also hopes to promote other various applications of the cell sheet regenerative medicine business in the United States. [1]-3 Japan: Collaboration with J-TEC
In January, CellSeed formed an "individual agreement for joint research and development" (Covering a three year period) for joint research of self cultured corneal epithelial layer with J-TEC. CellSeed' s cell sheet regenerative medicine technology will be applied to development of J-TEC' s self cultured corneal epithelial layers, and will be used in various aspects of cell sheet technologies including joint research and development. J-TEC will promote research and development of slightly modified variations of the self cultured corneal epithelial layer products in accordance with the "basic agreement for joint research and development," and submit safety approval applications of these products as quickly as possible.J-TEC was able to successfully launch the first regenerative medicine product within Japan as a commercial business, and is the only company in Japan with the capacity to produce regenerative medical products. Fujifilm Holdings Corporation is J-TEC' s main shareholder with 41% of total voting rights. [2] Patent Establishment
During the first half of the current term, patents for "epidermal cell sheet" and "high density cell array use substrate" have been acquired. These patents are a reflection of the novelty and progressiveness of cell sheet regenerative medical technologies, and these applications are gathering attention within the industry.
[2]-1 Transplant Use "Epidermal Cell Sheet" Patent: Japan
This patent is for the technology used to create tissue structure (Cultured epidermal cell sheets, cultured multiple layer epidermal sheets) from epidermal mucosal cells used in cell sheet regenerative medical technologies. This technology will enable a wide range of applications for reconstructed epidermal and mucosal related tissue structures (For example, corneal regeneration epithelial cell sheet and esophageal regeneration epithelial cell sheet.).
[2]-2 High Density Cell Array Use Substrate Patent: Japan
This is a patent for a new type of temperature responsive cell cultureware concept called "cell array use substrate." "Cell array use substrate" is a next generation technology of the "UpCell" series of cell sheet recovery use temperature responsive cell cultureware that allows the cultureware surface to be used in the gene delivery to cells and in the drug evaluation process. A characteristic of this process is its ability to uniformly align multiple cells. Therefore the temperature responsive cell cultureware principle enables users to target specific cells and separate and collect them without harming them.
|
| Fiscal Year December 2011 Earnings Estimates |
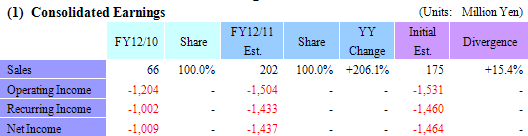 Full Year Earnings Estimates Remain Unchanged, Recurring Loss of ¥1.433 Billion Expected (¥1.002 Billion Recurring Loss in FY12/10)
Sales are expected to grow by 206.1% year-over-year to ¥202 million in fiscal year December 2011. Of this total, ¥68 million is expected to be derived from the regenerative medicine support business, and ¥134 million from the cell sheet regenerative medicine business. A lump sum payment of ¥127 million (Previously expected to be ¥107 million at the beginning of the term.) is expected to be derived from the individual agreement for the business launch of corneal regeneration epithelial cell sheet in the United States and ¥7 million from compassionate use of products in the cell sheet regenerative medicine business. With regards to profits, higher than expected sales of cell sheet regenerative medicine business is expected to allow profits to exceed expectations as well.
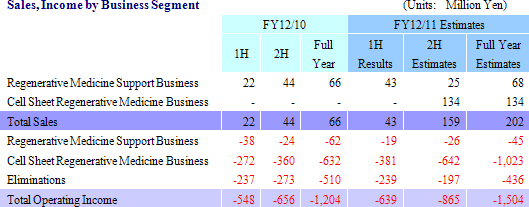 (2) Major Developments Anticipated from the Third Quarter Forward
In relation to corneal regeneration epithelial cell sheets, provision of products on a compassionate use basis are expected to begin and contribute ¥7 million to sales during the remainder of the term. Furthermore lump sum payment from the agreement with Emmaus Medical for joint research and development and commercialization in the United States is also expected. Aside from corneal regeneration epithelial cell sheet in the cell sheet regenerative medicine business pipeline, lump sum payment from the agreement for joint research and development is anticipated, in addition to completion of preparations for clinical research on esophageal regeneration epithelial cell sheet and regenerated cartilage sheet.
 |
| Conclusions |
|
Furthermore, cash flow is expected to improve dramatically due to the outlook for the lump sum payment of US$10 million expected to be received at the end of the current term. This improved cash flow will allow CellSeed to secure the funds necessary for the business development of its corneal regeneration epithelial cell sheet business in the United States and to conduct research and development of its other four product pipelines. Disclaimer
This report is intended solely for information purposes, and is not intended as a solicitation to invest in the shares of this company. The information and opinions contained within this report are based on data made publicly available by the Company, and comes from sources that we judge to be reliable. However we cannot guarantee the accuracy or completeness of the data. This report is not a guarantee of the accuracy, completeness or validity of said information and or opinions, nor do we bear any responsibility for the same. All rights pertaining to this report belong to Investment Bridge Co., Ltd., which may change the contents thereof at any time without prior notice. All investment decisions are the responsibility of the individual and should be made only after proper consideration.Copyright(C) 2011, All Rights Reserved by Investment Bridge Co., Ltd. |



